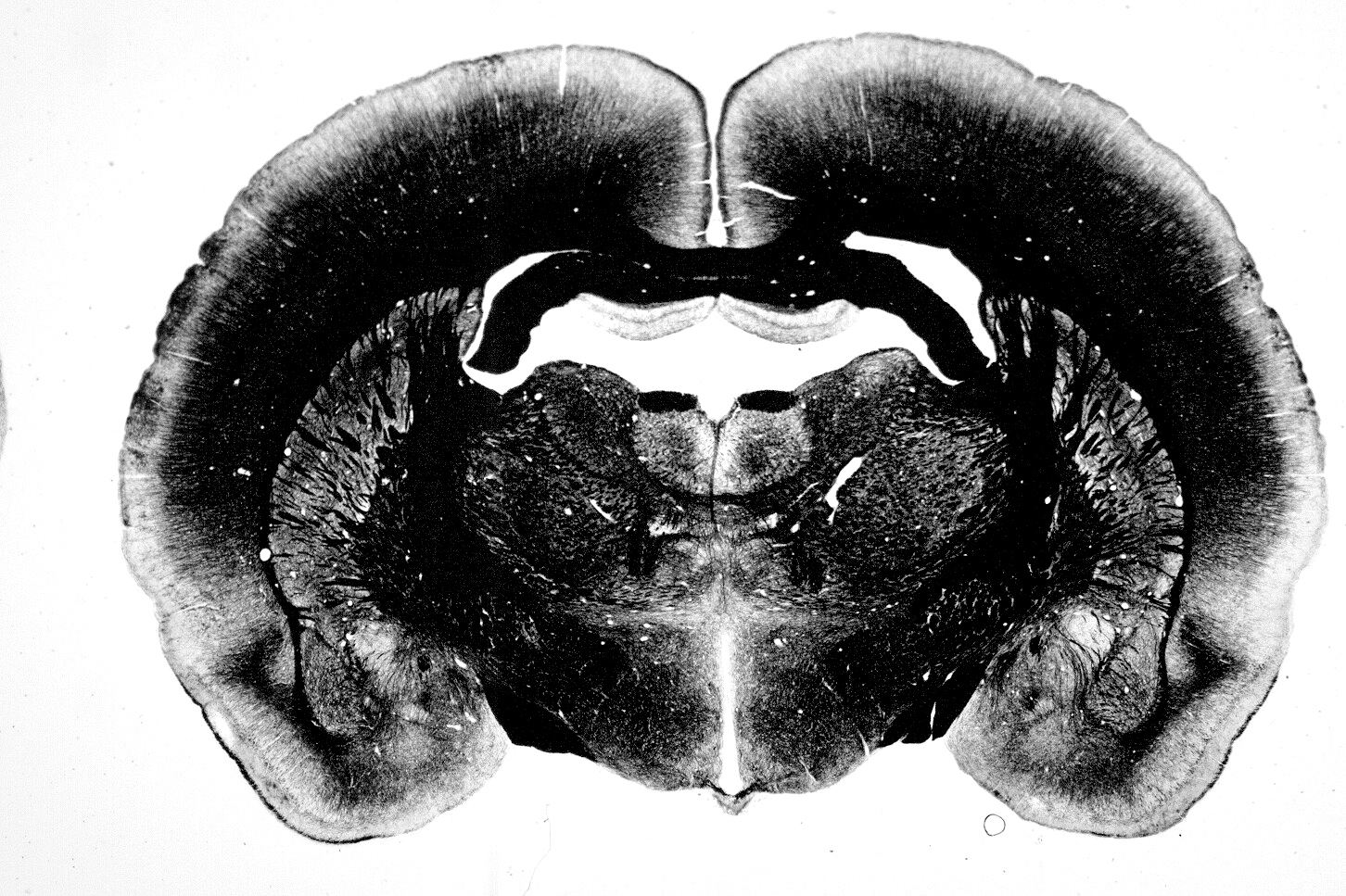
Our research focuses on how epigenetic mechanisms mediate the organizational effects of sex hormones on the brain during the critical perinatal period. In particular, I investigate how early exposure to testosterone regulates active DNA demethylation and DNA repair processes in the prefrontal cortex, and how these mechanisms contribute to the establishment of sex differences in gene expression associated with neurodevelopmental disorders.
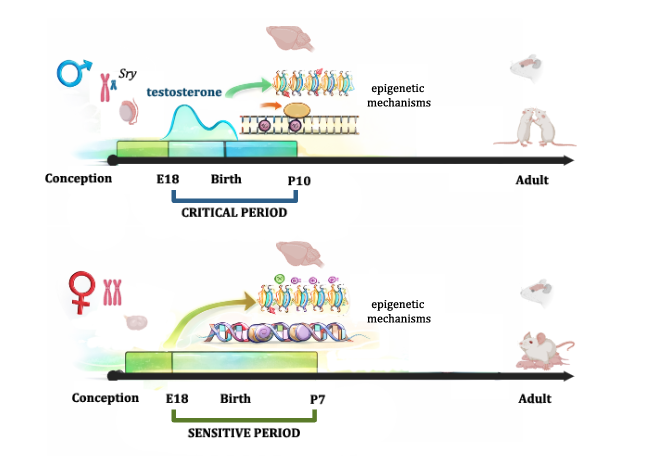
Several neuropsychiatric disorders, such as autism spectrum disorder and schizophrenia, originate during development and exhibit a sex bias in prevalence, being more common in males. These differences are largely linked to the transient action of gonadal hormones during a critical developmental window, whose organizational effects are long-lasting and generate a form of “cellular memory” in neural circuits.
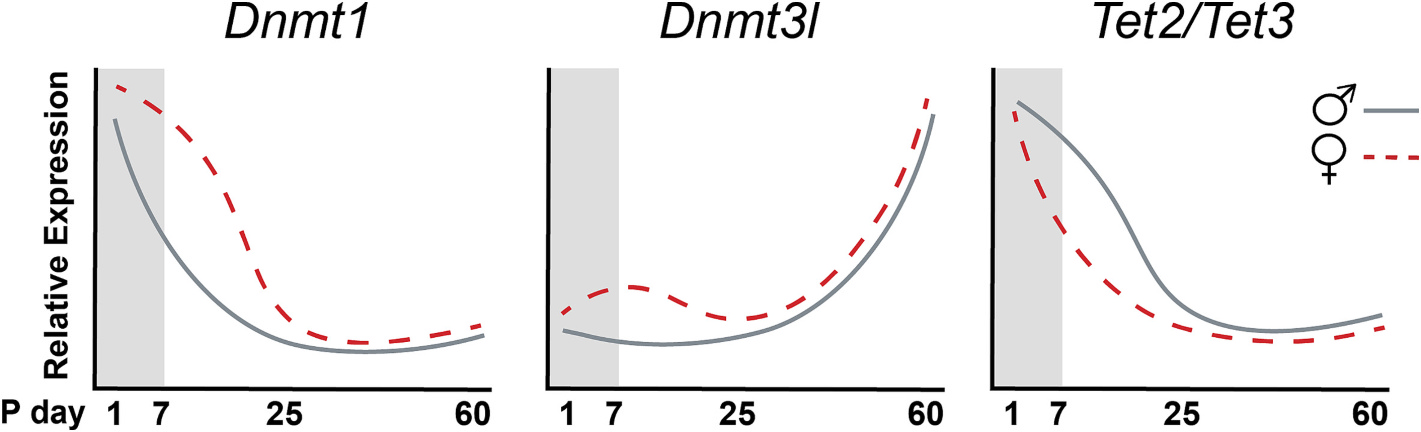
In recent years, DNA methylation has emerged as a key mechanism underlying these processes. Rather than being static, this epigenetic mark is highly dynamic in the developing brain, reflecting active demethylation pathways. In this context, enzymes of the TET family have been identified as central regulators of DNA demethylation, while also establishing hydroxymethylation as a functionally relevant epigenetic mark in the nervous system.
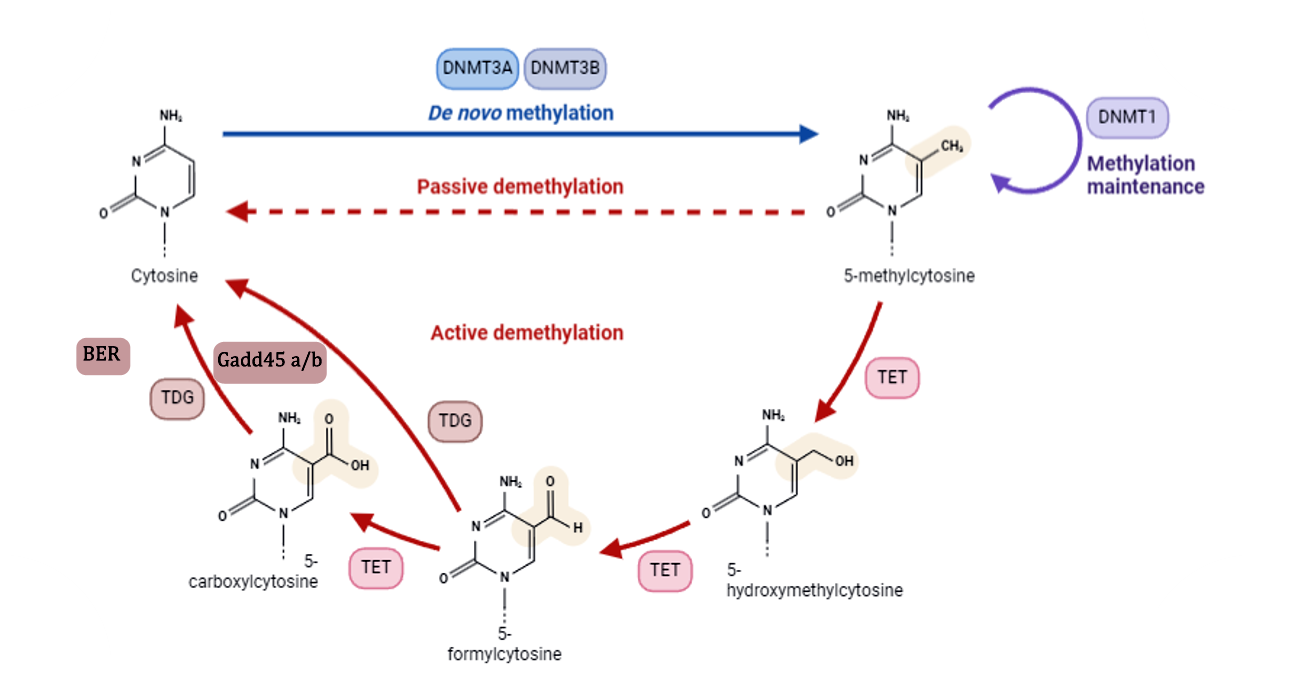
Our work proposes that, during development, the regulation of steroid-sensitive genes involves dynamic cycles of methylation and demethylation at promoter regions. Given that global levels of DNA methylation are lower in males during early development, we hypothesize that the balance between methylation and demethylation is shifted toward greater demethylation in males, creating a window of increased sensitivity to developmental perturbations.
In the long term, we aim to integrate epigenetic, neurobiological, and behavioral analyses from a sex-specific perspective to identify mechanisms underlying differential vulnerability and to uncover potential targets for prevention and therapeutic intervention in neurodevelopmental disorders.
COMPETITIVE RESEARCH FUNDS
- 2025. IBRO-Wellcome, Neuroscience Capacity Accelerator for Mental Health (NCAMH) Understanding Sex Differences in PTSD Vulnerability. Co-leader
- 2022-2024 PIBAA-CONICET. Rol de los esteroides gonadales en el control de la dinámica de metilación/demetilación del ADN durante el neurodesarrollo. PI
- 2022-2024. PICT-2020-SERIEA-00190. Fondo para la Investigación Científica y Tecnológica (FONCYT) Agencia Nacional de Promoción Científica y Tecnológica. PI.
- 2021. Research Supplies for use in the applicant’s home laboratory. Committee for Aid and Education in Neurochemistry (CAEN) – International Society for Neurochemistry (ISN). PI.
- 2019. Return Home Fellowship. International Brain Research Organization (IBRO). PI.
PUBLICATIONS (LATEST FIVE YEARS)
- Pasquetta L, Dimundo J, Cisternas CD, Piovano J, Miranda-Morales RS. Parental care shapes anxiety-like behavior, oxytocin, social interaction, and ethanol sensitivity in adolescent C57BL/6J mice. Pharmacol Biochem Behav. 2026;263:174188. doi:10.1016/j.pbb.2026.174188.
- Sosa C, Cabrera-Zapata LE, Cisternas CD, Arevalo MA, Cambiasso MJ. Sex chromosome-dependent epigenetic regulation underlies sex-specific H4 acetylation at the aromatase promoter in the developing mouse amygdala Biol Sex Differ. 2026;17(1):57. doi: 10.1186/s13293-026-00854-4.
- Castillo-Ruiz A, Cisternas CD, Sturgeon H, Forger NG. Birth triggers an inflammatory response in the neonatal periphery and brain. 2022 Brain Behav Immun. S0889-1591(22)00152-0. doi:10.1016/j.bbi.2022.05.021
- Cabrera Zapata LE, Cisternas CD, Sosa C, Garcia-Segura LM, Arevalo MA, Cambiasso MJ. X-linked histone H3K27 demethylase Kdm6a regulates sexually dimorphic differentiation of hypothalamic neurons. Cell Mol Life Sci. 2021 78(21-22):7043-7060. doi:10.1007/s00018-021-03945-0.
- Cortes LR, Cisternas CD, Cabahug INKV, Mason D, Ramlall EK, Castillo-Ruiz A, Forger NG. DNA methylation and demethylation underlie the sex difference in estrogen receptor alpha in the arcuate nucleus. Neuroendocrinology. 2021 doi:10.1159/000519671.
- Hoffiz YC, Castillo-Ruiz A, Hall MAL, Hite TA, Gray JM, Cisternas CD, Cortes LR, Jacobs AJ, Forger NG. Birth elicits a conserved neuroendocrine response with implications for perinatal osmoregulation and neuronal cell death. Sci Rep. 11(1):2335. 2021 doi:10.1038/s41598-021- 81511-1
- Cisternas CD, Cabrera Zapata LE, Mir FR, Scerbo MJ, Arevalo MA, Garcia-Segura LM, Cambiasso MJ. Estradiol-dependent axogenesis and Ngn3 expression are determined by XY sex chromosome complement in hypothalamic neurons. Sci Rep. 10(1):8223. 2020. doi: 10.1038/s41598-020-65183-x
- Cisternas CD, Cortes LR, Golynker I, Castillo-Ruiz A, Forger NG. Neonatal Inhibition of DNA Methylation Disrupts Testosterone-Dependent Masculinization of Neurochemical Phenotype. Endocrinology 161(1). 2020. doi: 10.1210/endocr/bqz022.
- Cisternas CD, Cortes LR, Bruggeman E, Yao B, Forger NG. Developmental changes and sex differences in the expression of DNA methylation and demethylation enzymes in hypothalamic regions of the mouse brain. Epigenetics, 15 (1-2), 72-84. 2020. doi: 10.1080/15592294.2019.1649528.
- Jacobs A, Castillo-Ruiz A, Cisternas CD, Forger NG. Microglial depletion causes region-specific effects on developmental neuronal cell death in the mouse brain. Developmental Neurobiology. 2019. doi:10.1002/dneu.22706.
- Cortes LR*, Cisternas CD*, Forger NG. Does gender leave an epigenetic imprint on the brain? Frontiers in Neuroscience. 13:173. 2019 doi:10.3389/fnins.2019.00173. Review. *co-first authors.
- Complete and updated list available here.
TEAM